Scientific Calendar September 2020
Thrombotic thrombocytopenic purpura (TTP)
What are the laboratory findings in patients with thrombotic thrombocytopenic purpura (TTP)?
Platelet counts > 30 x 109/L, LDH increased, haemoglobin and haptoglobin decreased, schistocytes on peripheral blood smear, ADAMTS13 concentration 20 – 40 % of the norm, and normal presentation of VWF.
Platelet counts > 30 x 109/L, LDH, haemoglobin and haptoglobin decreased, peripheral blood smear with no conspicuous findings, ADAMTS13 concentration < 10 % of the norm, and unusually large VWF multimers.
Platelet counts < 30 x 109/L, increased LDH, haemoglobin and haptoglobin decreased, schistocytes on peripheral blood smear, ADAMTS13 concentration < 10 % of the norm, and unusually large VWF multimers.
Congratulations!
That's the correct answer!
Sorry! That´s not completely correct!
Please try again
Sorry! That's not the correct answer!
Please try again
Notice
Please select at least one answer
Scientific background
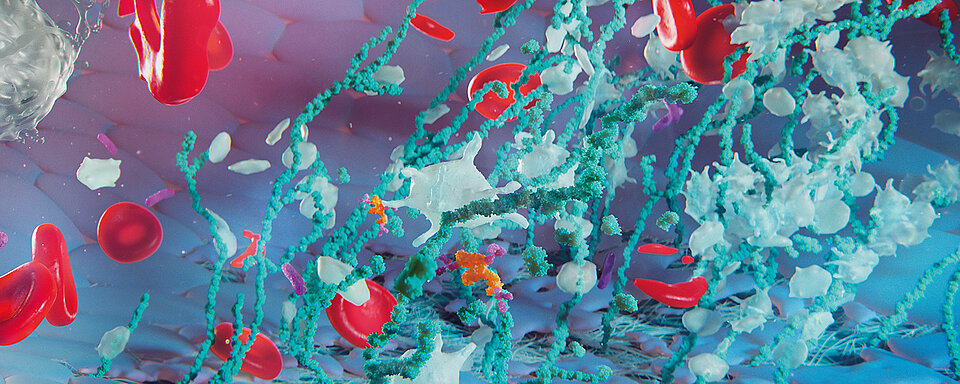
Thrombotic thrombocytopenic purpura (TTP) is a blood disorder and defined by the presence of microangiopathic haemolytic anaemia (MAHA) and moderate to severe thrombocytopenia, purpura or bleeding associated with thrombocytopenia, fever, neurological impairment and renal disease presenting with haematuria and/or proteinuria or elevated blood urea nitrogen.
Laboratory findings of platelet counts usually below 30 x 109/L, increased lactate dehydrogenase [LDH] levels, reduced haemoglobin and haptoglobin concentrations, the presence of schistocytes on the peripheral blood smear, ‘unusually large’ (UL) multimers of von Willebrand factor (VWF), and plasma concentrations of ‘A Disintegrin And Metalloproteinase with a ThromboSpondin type 1 motif, member 13’ (ADAMTS13) of below 10 % of the norm are highly specific for TTP in the presence of clinical features [1, 2].
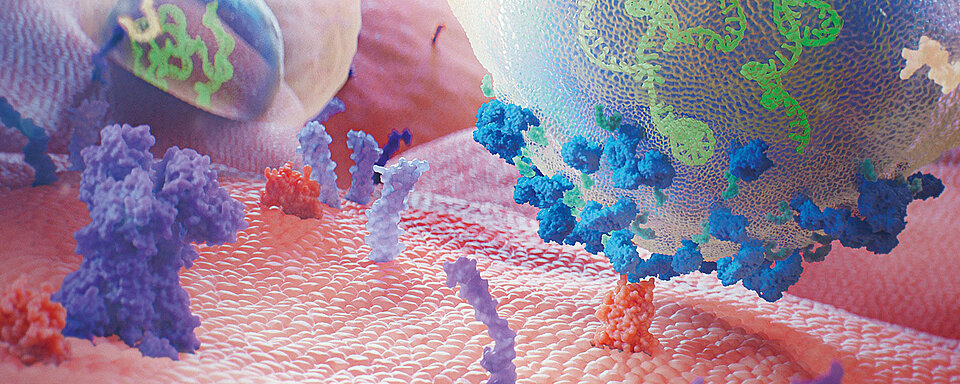
The specific VWF-cleaving protease ADAMTS13 regulates the VWF multimers after secretion from the endothelial cells. It cleaves the Y1605-M1606 bond in the A2 domain. Under high shear forces, the VWF multimers secreted from the endothelial cells remain bound to the endothelium and are sustained by the shear stress. This makes the cleavage sites in the A2 domains accessible for the freely circulating ADAMTS13 to cleave the VWF multimers. Under normal, non-thrombotic conditions, the remaining VWF is not reactive. Due to its property as an acute phase protein, infections or inflammatory processes may lead to an increased release of VWF. In individuals with acquired or congenital ADAMTS13 deficiency, this can lead to the formation of microthrombi and the development of clinical signs of a TTP [3, 4].
It is important to distinguish TTP from other thrombotic microangiopathies (TMA). In patients with infection-associated TMA, MAHA and thrombocytopenia can develop due to systemic bacterial, viral or mycotic infections. Therefore, infection-associated TMA should always be considered during the evaluation of such patients, particularly elderly patients who have fever or chills associated with features of TTP. An infection can also trigger an acute episode of TTP, and it may be difficult to differentiate whether the infection is the cause or a trigger [5]. The ADAMTS-13 activity in patients with infection-associated TMA is usually discretely reduced to 20 % of the norm or even higher, with some exceptions in patients with e.g. severe sepsis causing a severe ADAMTS-13 deficiency in the absence of an inhibitor [6, 8]. Infection-associated TMA is triggered by endothelial damage caused by infection leading to increased VWF release and increased consumption of ADAMTS-13. Bacterial enzymes and cytokines lead to ADAMTS-13 proteolysis and inhibition [6]. Additionally, neutrophil extracellular traps (NETs) – chromatin fibres released from neutrophils during infection – may result in thrombosis because of their ability to cause platelet activation [3, 4, 7]. In patients positive for human immunodeficiency viruses (HIV), features of TMA may be due to multiple causes including coexisting TTP. These include advanced disease with opportunistic infections, malignancies and antiviral medications. In addition, the initiation of highly active antiretroviral therapy (HAART) may inhibit ADAMTS-13 activity and result in features of TMA [9, 10].
References
[1] Zheng XL, Kaufman RM, Goodnough LT, Sadler JE (2004): Effect of plasma exchange on plasma ADAMTS13 metalloprotease activity, inhibitor level, and clinical outcome in patients with idiopathic and non-idiopathic thrombotic thrombocytopenic purpura. Blood; 103: 4043–9.
[2] Rock GA, Shumak KH, Buskard NA, Blanchette VS, Kelton JG, Nair RC, Spasoff RA (1991): Comparison of plasma exchange with plasma infusion in the treatment of thrombotic thrombocytopenic purpura. Canadian Apheresis Study Group. N Engl J Med; 325: 393–7.
[3] Kalagara T, Moutsis T, Yang Y, et al. (2018): The endothelial glycocalyx anchors von Willebrand factor fibers to the vascular endothelium. Blood Adv.; 2(18):2347–57. doi:10.1182/bloodadvances.2017013995
[4] Nazy I, Elliott TD and Arnold DM (2020): Platelet factor 4 inhibits ADAMTS13 activity and regulates the multimeric distribution of von Willebrand factor. Br J Haematol. doi:10.1111/bjh.16553
[5] Saha M, McDaniel JK, Zheng XL (2017): Thrombotic thrombocytopenic purpura: pathogenesis, diagnosis and potential novel therapeutics. J Thromb Haemost; 15: 1889–900.
[6] Schwameis M, Schorgenhofer C, Assinger A, Steiner MM, Jilma B (2015): VWF excess and ADAMTS13 deficiency: a unifying pathomechanism linking inflammation to thrombosis in DIC, malaria, and TTP. Thromb Haemost; 113: 708–18.
[7] Ono T, Mimuro J, Madoiwa S, Soejima K, Kashiwakura Y, Ishiwata A, Takano K, Ohmori T, Sakata Y (2006): Severe secondary deficiency of von Willebrand factor-cleaving protease (ADAMTS13) in patients with sepsis-induced disseminated intravascular coagulation: its correlation with development of renal failure. Blood; 107: 528–34.
[8] Sorvillo et al. (2019): Plasma Peptidylarginine Deiminase IV Promotes VWF-Platelet String Formation and Accelerates Thrombosis After Vessel Injury. Circulation Research; 125(5): 507–19. https://doi.org/10.1161/CIRCRESAHA.118.314571
[9] George JN, Terrell DR, Vesely SK, Kremer Hovinga JA, Lammle B (2012): Thrombotic microangiopathic syndromes associated with drugs, HIV infection, hematopoietic stem cell transplantation and cancer. Presse Med; 41: e177–88.
[10] Malak S, Wolf M, Millot GA, Mariotte E, Veyradier A, Meynard JL, Korach JM, Malot S, Bussel A, Azoulay E, Boulanger E, Galicier L, Devaux E, Eschwege V, Gallien S, Adrie C, Schlemmer B, Rondeau E, Coppo P; Réseau d’Etude des Microangiopathies Thrombotiques (2008): Human immunodeficiency virus-associated thrombotic microangiopathies: clinical characteristics and outcome according to ADAMTS13 activity. Scand J Immunol; 68: 337–44.