MammaTyper®
Comprehensive molecular subtyping of breast cancer patients
- Precise determination of HER2, ER, PR and Ki-67 in one assay
- Definitive breast cancer subtyping for confident treatment decisions
- Easy-to-use, quantitative and observer-independent testing
- Fast turnaround time and results within the same day
- CE-marked IVD RT-qPCR assay for most commonly available cyclers
MammaTyper® precisely quantifies expression of HER2, ER, PR and Ki-67 mRNA
MammaTyper® is a molecular diagnostic test for quantitative determination of the four key biomarkers used in the subtyping of breast cancer.
Human epidermal growth factor receptor 2 (HER2), estrogen receptor (ER), progesterone receptor (PR) and the marker of proliferation Ki-67 are key biomarkers in the evaluation of breast cancer tumours.
The combination of biomarker results allows the assessment of the different St. Gallen Breast Cancer subtypes which are key parameters for treatment decisions.
Definition of Breast Cancer Surrogate Subtypes
(St Gallen 2013)
| Breast Cancer Subtypes | ER | PR | HER2 | Ki-67 |
| Luminal A-like | Pos | Pos | Neg | Neg |
| Luminal B-like (HER2 negative) | Pos | Pos/Neg* | Neg | Pos/Neg* |
| Luminal B-like (HER2 positive) | Pos | Pos/Neg | Pos | Pos/Neg |
| HER2 positive (non-luminal) | Neg | Neg | Pos | Pos/Neg |
| Triple negative (ductal) | Neg | Neg | Neg | Pos/Neg |
*with the exception of the combination PR pos and Ki-67 neg = Luminal A-like
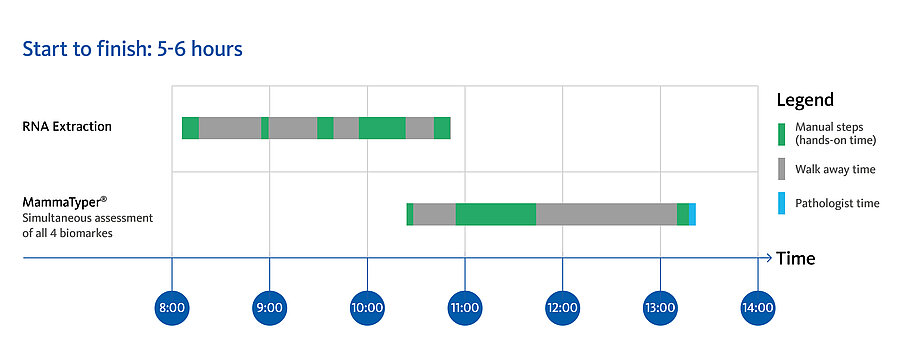
MammaTyper® is designed to meet the need for standardized, quantitative and fast molecular subtyping enabling confident treatment decisions.
MammaTyper® is an easy-to-use test for any pathology laboratory, delivering precise results within 6 hours.
| 1 | Sample preparation | 10 μm FFPE tissue section (tumor cell content > 20 %). |
| 2 | RNA extraction | Use of RNA FFPE sample extraction kit according to IFU or validated commercial RNA extraction systems is recommended. |
| 3 | MammaTyper® test set up | Preparation of mastermixes and distribution on 96 well plate. Analysis of up to 8 patient samples per run. |
| 4 | RT-qPCR analysis | Validated on the following qPCR instruments:
|
| 5 | Data processing and reporting | Convenient MammaTyper® Report Generator software:
|
MammaTyper® is a trademark in various jurisdictions, which is exclusively licensed to Cerca Biotech www.cercabiotech.com
| Feature | MammaTyper® IVD test kit (10 reactions) |
| Sample type | 10 μm FFPE tissue section (tumor cell content > 20 %) |
| Sample capacity | Up to 8 patient samples per kit |
| QC function | 2 external controls (positive + negative) |
| Compatiple platforms | LightCycler® 480 Instrument II (Roche) Cobas z® 480 Analyzer (Roche) Versant® kPCR AD module (Siemens) Applied Biosystems® 7500 Fast (Dx) RT PCR (ThermoFisher Scientific) CFX96TM RT PCR Detection System-IVD (BIO-RAD®) MX3000P (Agilent®) Agilent® AriaDx Real-Time PCR System (Agilent®) SLAN®-96P Real-Time PCR System (Sanure Biotech) |
| Concordance | Concordance between MammaTyper® and IHC/CISH-based biomarker assessments: HER2 91.8%, ER 91.8%, PR 82.5%, KI67 75.0% 1 Overall Percentage Agreement (OPA) of assessment by MammaTyper® vs IHC/FISH (Ki67 digital image analysis): HER2 95.0%, ER 95.5%, PR 89,4%, Ki67 87,2% 4 |
| Reproducibility | Reproducibility of binary single-marker results (pos/neg), as well as the molecular subtype agreement, was almost perfect (kappa values: 0,90 – 1,00) in a 10 centre concordance study 2 |
| Reclassification | MammaTyper® may offer more precise assessment of endocrine responsiveness, improve Ki67 standardization and help resolve eqivocal HER2 IHC/FISH cases, leading to potential redistributions of the molecular subtypes 3 |
| Catalogue Number | CC01010 |
References
- Wirtz RM et al., Breast Cancer Res Treat 2016; 157(3), 437446
- Varga Z et al., Breast Cancer Res 2017; 19(55), 1-13
- Caselli E et al., PLOS ONE 2021; 16(9), 1-18
- Shaaban A et al., Eur J Cancer 2022; 175(1), 87-88
Sysmex UK LTD
Sysmex House
Garamonde Drive
Wymbush
Milton Keynes
MK8 8DF
0333 320 3460
Product documents
Regulatory Documents
Regulatory documents, such as Instructions for Use, can be accessed with a valid My Sysmex login:
Go to My Sysmex![[.CO.UK-en United Kingdom (english)] MammaTyper Box](/fileadmin/_processed_/a/d/csm_MammaTyper_3_b15f9fc541.png)
![[.CO.UK-en United Kingdom (english)] MammaTyper Vials](/fileadmin/_processed_/6/8/csm_MammaTyper_2_022fedd638.png)

![[.CO.UK-en United Kingdom (english)] MammaTyper Box](/fileadmin/media/f100/Products/MammaTyper_3.png)
![[.CO.UK-en United Kingdom (english)] MammaTyper Vials](/fileadmin/media/f100/Products/MammaTyper_2.png)